What are Exosomes?
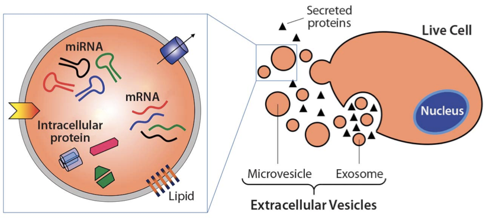
Exosomes are nanosized, extracellular vesicles secreted by a variety of cells, including stem cells. They carry important signals from one cell to another in the form of “cargoes,” such as microRNAs or messenger RNAs. These can induce changes in the behavior of the recipient cells, including reducing a diseased cell behavior or “phenotype,” including inflammation.
Exosome treatments circumvent many of the challenges and concerns associated with stem cell transplantation (immunorejection, tumorigenesis, limited survival/assimilation).
They allow for selective control over what contents are delivered my measuring or modifying their cargoes.
Exosomes are blood-brain barrier permeable and can migrate between cells in ways that implanted stem cells can’t. This allows for unique advantages in drug delivery.
Exosomes can be engineered to target specific cell types.
01-22-Das-lab-Exosomes-article-figure1.png
From Wikipedia: “Exosomes are cell-derived vesicles that are present in many and perhaps all biological fluids, including blood, urine, and cultured medium of cell cultures. Exosomes contain various molecular constituents of their cell of origin, including proteins and RNA. It is becoming increasingly clear that exosomes have specialized functions and play a key role in, for example, coagulation, intercellular signaling, and waste management.”
Stem Cell Isolation and Preservation Process
Stem Cell Isolation from Umbilical Cords (UCs), one of several sources of the stem cells being researched at StemXo:
| 1) UCs are washed and incubated in low-glucose growth medium 2) UCs are washed again and injected with collagenase and low-glucose growth medium 3) Tissue is massaged and cells are harvested from solution 4) Cells are isolated and cultured in culture medium | 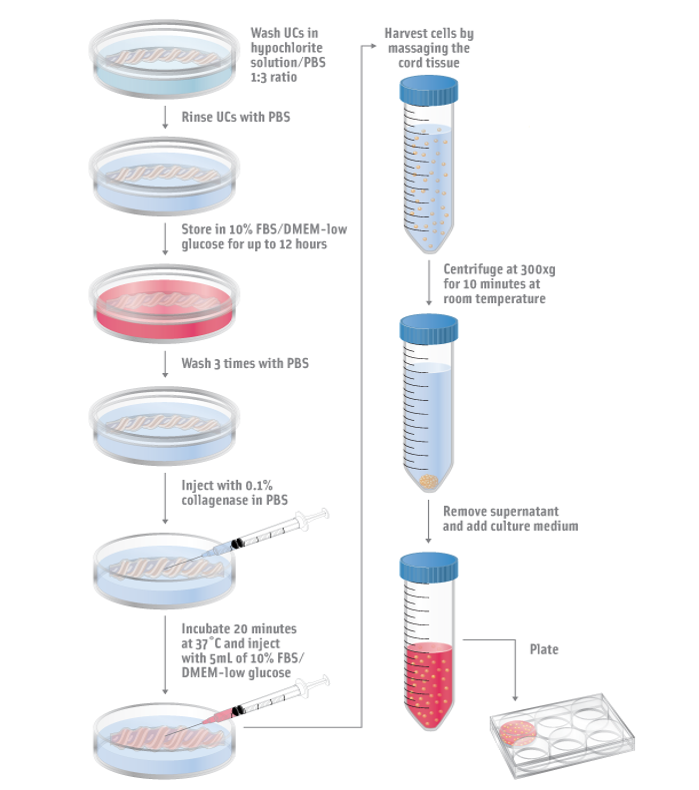 |
Exosome Isolation Process from Stem Cells
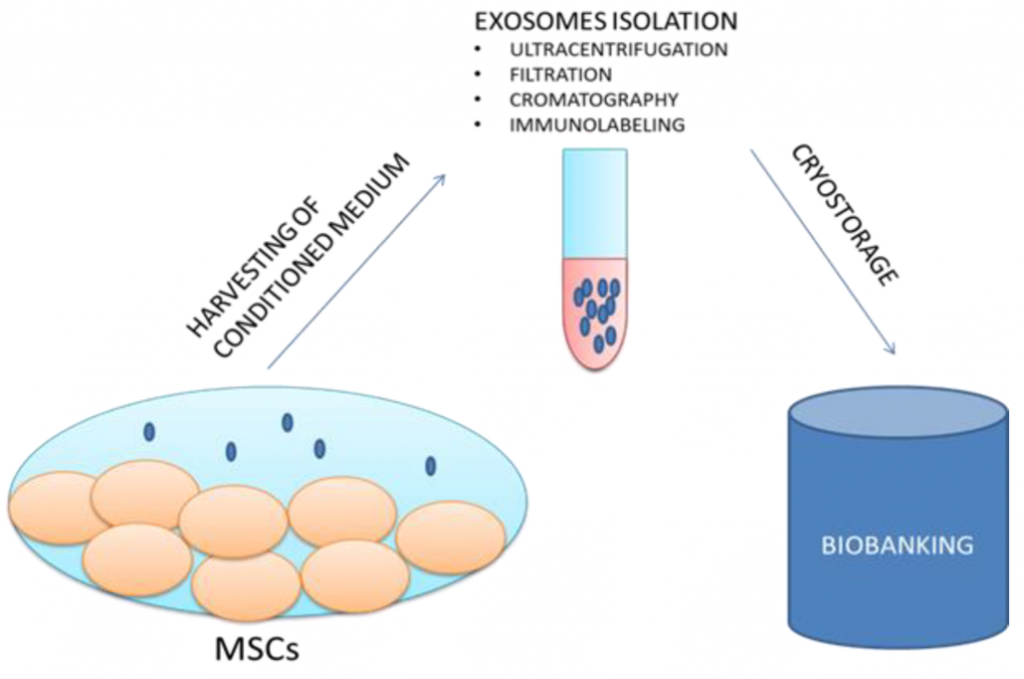
Exosome isolation from human stem cells:
- Stem cells are cultured in sterile plate
- Secreted exosomes are harvested in conditioned medium
- Exosomes are isolated and purified
- Exosomes are stored for preservation
Additional StemXO Research Interests
iPSC-based Platform Development of Cell Lineage-Specific Therapies
- Human induced pluripotent stem cells (iPSCs) are cells derived from human somatic tissues such as skin fibroblasts, which are then reprogrammed into stem cells
- iPSCs can be differentiated into a wide variety of cell types, each with their own unique properties and therapeutic potential. As such, they have incredible potential as a platform for the development of multiple therapies for human disease
- iPSCs can also be used to model disease states. If they are derived from patients with a known mutation or if a mutation is induced, the properties of specific cell types in the body derived from the parent iPSCs can then be explores to see how the disease affects them
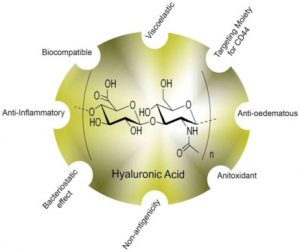
Hyaluronic acid (HA): a nano-carrier for exosome delivery
- Is a naturally-occurring macromolecule component of the brain extracellular matrix
- Is being studied as a gel matrix for stem cell delivery, tissue engineering, and brain repair after stroke
- Has been used experimentally to increase the stability and solubility of cancer drugs
- As a nanoemulsion, can be used to protect therapeutic exosomes for gradual release from delivery site at the nasal mucosa, into the brain
(Sources for images: Zhu Z, Yin-Min W, Jun Y. Hyaluronic acid: a versatile biomaterial in tissue engineering. Plast Aesthet Res. 2017;4(219-27)
Wickens JM, Alsaab HO, Kesharwani P, Bhise K, Amin MC, Tekade RK, Gupta U, Iyer AK. Recent advances in hyaluronic acid-decorated nanocarriers for targeted cancer therapy. Drug discovery today. 2017 Apr 1;22(4):665-80.
Shinde RL, Bharkad GP, Devarajan PV. Intranasal microemulsion for targeted nose to brain delivery in neurocysticercosis: role of docosahexaenoic acid. European Journal of Pharmaceutics and Biopharmaceutics. 2015 Oct 1;96:363-79.)
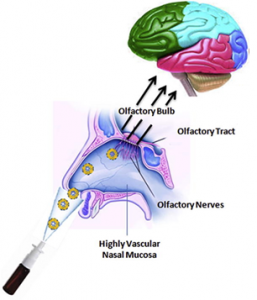

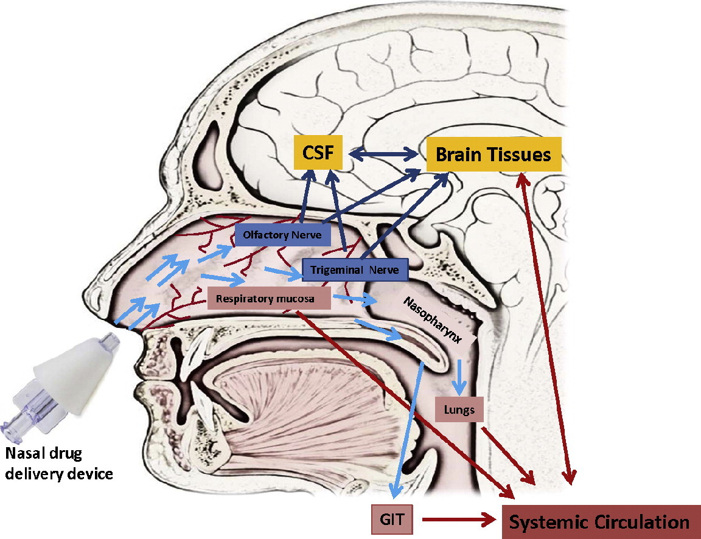
Intranasal Delivery of Therapeutics for CNS Pathologies
- Intranasal delivery involves delivering a therapeutic agent through the nostrils into the nasal cavity, for uptake into the brain
- This approach carries several advantages for enhanced delivery, as well as avoiding effects in non-brain organs when delivery to these (as through the bloodstream) is not desired
- This approach allows delivery in a way that is less invasive for many patients, and more convenient for at-home delivery
- Several components can be added to enhance nose-to-brain uptake
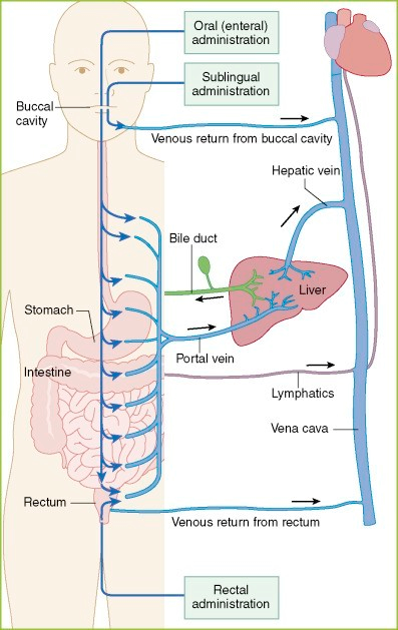
Biomaterial-assisted Sublingual Delivery of Therapeutics
- Biomaterial film matrices are being developed that can accommodate delivery of therapeutic agents through the sublingual/buccal route, under the tongue
- Sublingual delivery avoids first-pass metabolism through the gut and liver, providing more direct delivery to the bloodstream than oral administration
- This approach allows delivery to the bloodstream in a way that is less invasive for many patients than intravenous injection, and more convenient for at-home delivery